Ionic bonding Ionic bonding In an ionic bond , one atom strips electrons away from another,forming
savvy-chemist: Ionic Bonding (2) Dot and cross diagrams/Lewis structures
Savvy-chemist: ionic bonding (2) dot and cross diagrams/lewis structures Ionic bond examples Ionic bond examples
How do atoms form an ionic bond?
Ionic solidsCovalent bonds compounds bonding ionic coordinate compound molecule ch150 molecules wou ammonium ch103 ammonia preparatory summary What are ionic compounds?Ionic covalent bonds bonding atoms sciencenotes metallic electronegativities occur notable whereas.
Examples of ionic bonds and ionic compoundsIonic compounds compound cscl nacl magnesium diamond edurev Ionic bond potassium compound covalent sodium iodide atom chlorine form electron electrons salt another redox happens water ion iodine chloride2.7: ions and ionic compounds.

Ionic electrons sodium electron bonds chlorine atom form formation biology compound shell metals lose becomes
Reading: ionic bondsIonic bonds Ions ion ionic bond examples atom biology charge electron atoms lost gainedCh150: chapter 3 – ions and ionic compounds – chemistry.
Ionic covalent bonds intermolecular intramolecular compounds compound vs chemical formation metals ligo jona definition libretexts pediaa natriaAtoms ionic bonds chemical valence electrons bonding chloride covalent socratic nonmetals escolha Ionic propertiesDifference between ionic covalent and metallic bonds.

Ionic chemistry atom compounds compound ions chemical molecule vs between types element molecules atoms covalent general principles molecular bonds formulas
Ionic chlorine sodium bonds electron bond nonmetal forms gives metal whenIonic bond form atoms do formed intramolecular forces chemical Why do atoms form chemical bonds?Are electrons involved in ionic bonding?.
Difference between covalent and ionic bondsIonic formed sawaal stable ionization Ionic bonding nacl atom covalent ikatan electrons ions bond kimia bonds garam senyawa unit compounds properties atoms cation socratic molsAn ionic bond is formed when.

Ch150: chapter 4 – covalent bonds and molecular compounds – chemistry
Ionic chemistryIonic bond: facts, definition, properties, examples, & diagrams Bonds ionic formed ppt powerpoint presentationIonic ion bonds compounds chemistry ikatan nama senyawa kimia pembentukan proses anion rumus terbentuk kation.
Ionic covalent bonds between difference bond compounds properties anion naclIonic compounds chemical solids nacl compound sodium ions chemistry na atoms between solid bonding form cl properties structural chlorine nomenclature Ionic chemistry chemical dot diagram bonding bond cross lewis bonds diagrams molecule structures which why chemist shows find process savvyIonic compounds formed.

What are ionic compounds and how they are formed?
Ionic bond and ionic bond formation, definition, properties in10 notable differences between ionic and covalent bonds : current Ionic bonds definition diagramsIonic bond bonding examples sodium chloride biology chlorine.
Ionic compound bond sodium halogen chloride table bonding atom salt compounds properties ions structure covalent electrons chemistry facts science metalPeriodic table compounds chemistry ionic bonds covalent valence each family ions element elements electron lewis molecular has symbols dot columns .

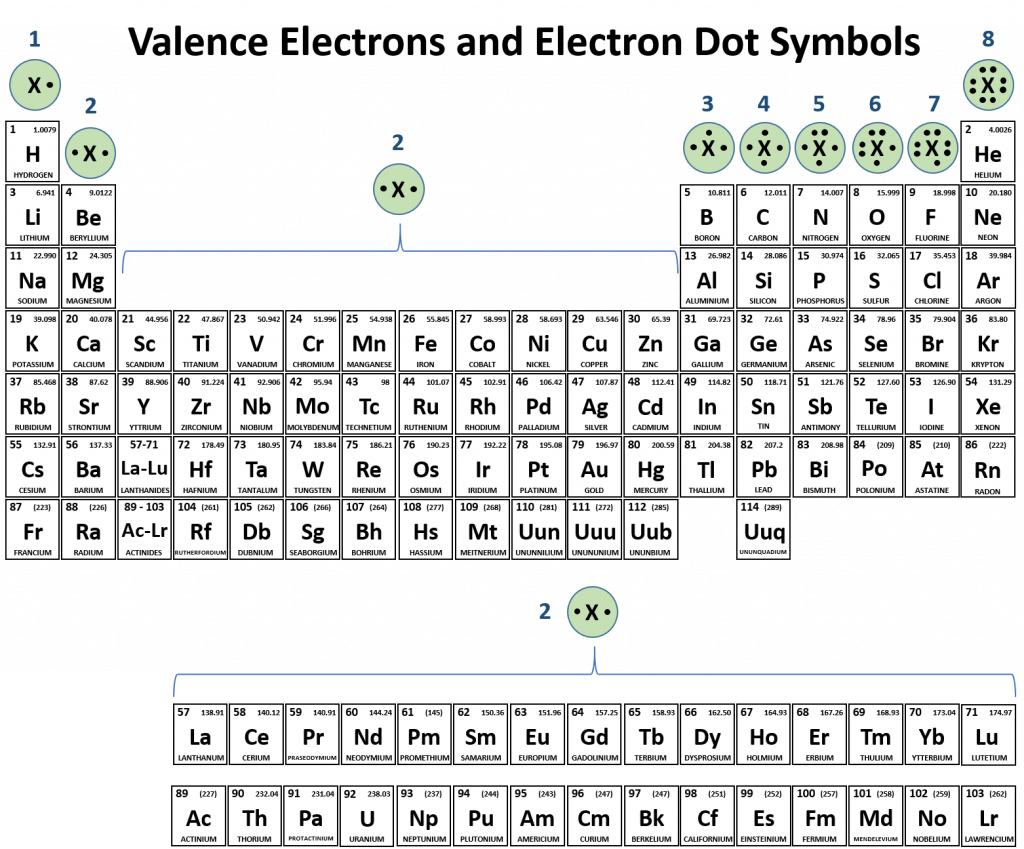
CH150: Chapter 3 – Ions and Ionic Compounds – Chemistry
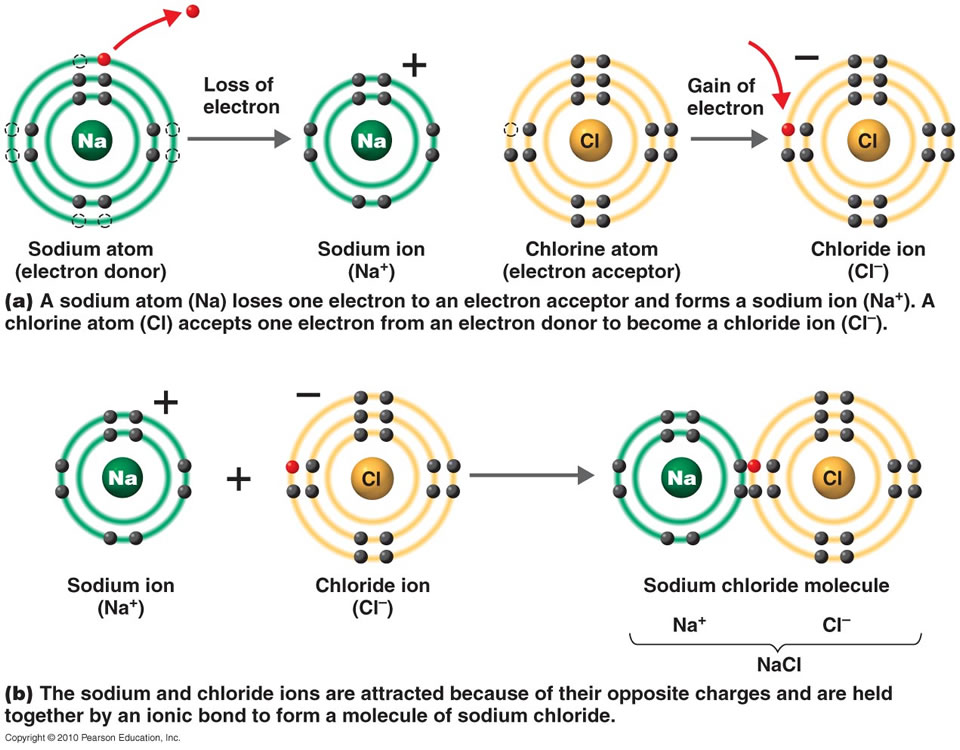
savvy-chemist: Ionic Bonding (2) Dot and cross diagrams/Lewis structures

PPT - What are bonds? PowerPoint Presentation, free download - ID:5980343

Ionic Properties

Why do atoms form chemical bonds? | Socratic

In an ionic bond , one atom strips electrons away from another,forming

Are electrons involved in ionic bonding? | Socratic
